Applications of the Schrödinger Equation
Sunday, February 27, 2022
Constant Potential Energy
If the potential energy is constant, the Schrödinger equation becomes:
Assuming
Then using the fact that
from the equation for the wave function, , the wave number, , can be represented as
The solution to this equation gives a form where the second derivative is equal to itself multiplied by a negative constant, meaning it can be represented in the form
If (forbidden region)
Rewriting the solution for the case where , we get
where
In this case, the solution can generally be written as
The Free Particle
For any free particle, the force it experiences is 0. This means the potential energy is constant (we say ), so the energy of the particle is
using the form . Notice how can have any value, meaning the energy is not quantized.
Normalization integral
Since this cannot be integrated from to , we cannot normalize the wave function in this form. Instead, we use the complex exponentials:
where
The interpretation of this solution relies on the time-dependent version of the Schrödinger equation:
The first term represents a wave moving to the right with amplitude , whereas the second term represents a wave moving to the left with amplitude .
Beam of particles
To represent a beam of particles moving in the direction, set . Then, the probability density function becomes
which makes sense because the particles are equally likely to be found anywhere along the axis, giving a completely unlocalized wave.
Infinite Potential Energy Well
Setup
The particle is trapped in a region of width between two infinitely high walls of infinite potential energy. It makes perfectly elastic collisions with either side, so its energy is completely confined to the region. It is said that
Outside the region
To find the solution outside the region, it is quite clear that must be 0 so that is not infinite (since ). Additionally, if the particle is always contained in the region, the probability of finding it outside the region must be .
Inside the region
Knowing inside the region, the solution becomes
where
To ensure the equation for the wave is continuous at the boundaries where and , we set the following restrictions:
The first of these equations gives , leaving . Either (which would mean the wave is 0 everywhere) or , which is only true when
Solving for the wavelength, we find that .
The solution to the Schrödinger equation for a particle trapped in a linear region is a series of standing de Broglie waves
And since only certain wavelengths are permitted, we also can see that
The energy is quantized as well
Allowed energies
Setting , we find that . Therefore, only square integer multiples of are allowed. , when , is called the ground state, and all other values of are called the excited states.
By absorbing or releasing energy equal to the difference in energy states, the particle can transition between one excited state and another.
Normalization
The final step in the solution for is to determine the value of that normalizes the function:
Solving this makes , so
Between two points, the wave function and probability distributions look something like this for the first few values of :
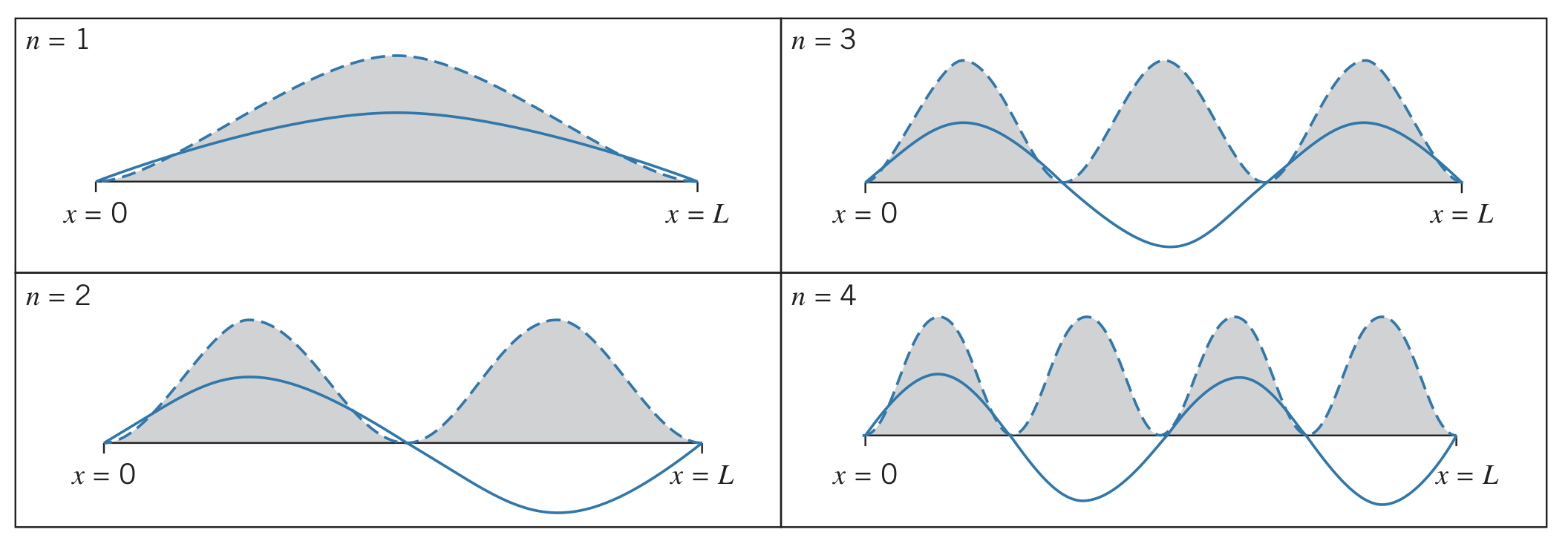
This is very strange behavior since it means a particle can go between two locations in space without going in the spaces in between them.
Motion of the particle
For this trapped particle, the uncertainty in position would be and the uncertainty in momentum would be . Therefore, the product of these two uncertainties is
This means the product of the uncertainties grows as increases. The minimum value occurs when :
Finite Potential Energy Well
A finite potential energy well can be defined as such:
Inside the region
Inside the region is the same as it was for an infinite energy potential well:
although the values for and are not valid in this calculation.
For the region
In this case, the energy of the particle is less than its potential energy, so we must use the form . However, since the region includes , the second term would become infinite. Therefore, and the equation becomes
For the region
In this region, we must again use the form . However, since this region includes , . Therefore,
Solving
We now have five unknowns: coefficients , , , , and the energy, . Additionally, we have four equations from the boundary conditions (continuity for and now at both and ) and one equation from the normalization condition.
The energy values must be determined numerically given the situation since the resulting equation results in a transcendental equation that cannot be solved algebraically.
These equations can be solved with specific numbers, given a problem. The key quality of any solution however is that at the boundary conditions, the wave function satisfies both the continuity of the function itself and the continuity of the first derivative of the function, such as the examples shown below:
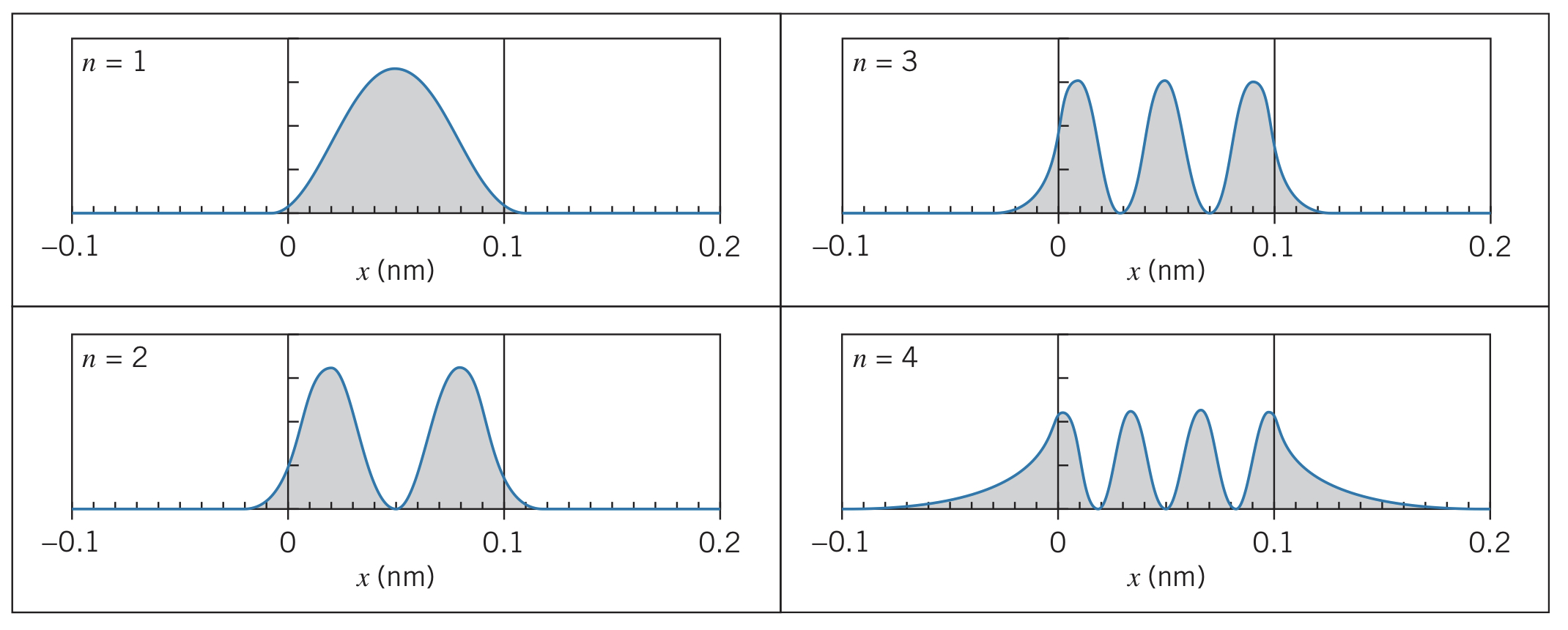
In these graphs, the penetration property is also clearly seen, as the sinusoidal functions transition into exponential ones at the boundaries. Note that for higher energy levels, the wave penetrates further into the forbidden region.
Two-Dimensional Infinite Potential Energy Well
Extending the Schrödinger equation to two dimensions, we get:
And the two-dimensional version of the infinite potential energy well is defined as follows:
This represents a particle confined in an square, where the corners are , , , and . Collisions with the lines , , , and are perfectly elastic.
Just like the previous one-dimensional wells, we assume the function is 0 in the forbidden regions. But now, we also assume the wave function is separable in the well. That is,
where
Applying the boundary conditions, we see that
Using these conditions, we see that , just like the one-dimensional case. We also see that since and , and must be integer multiples of (but not necessarily the same multiple).
Putting it all together, we see that
We can find coefficient using the normalization of the function:
Or for this situation,
Solving this equation gives us that
Energy
Plugging this solution into the energy equation, we find that the allowed energies for values of and are
The ground state for this situation occurs when :
Therefore,
and are called quantum numbers, and different combinations of each of them produce different probability density distributions. Just like the one dimensional case, where the value of is highest, we expect to find the particle and where it is 0, we do not expect to find the particle. See the examples below:
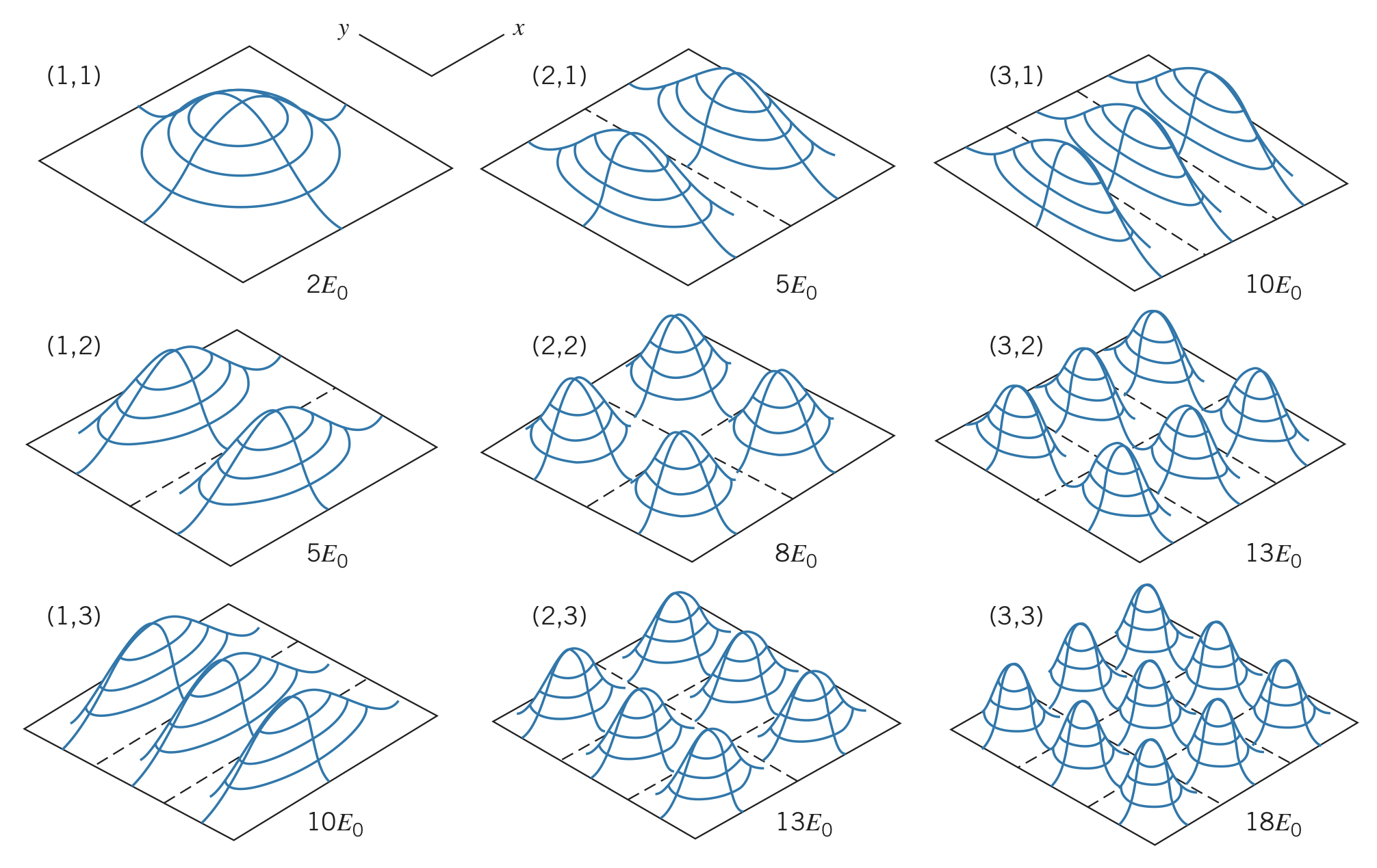
Experimental accuracy
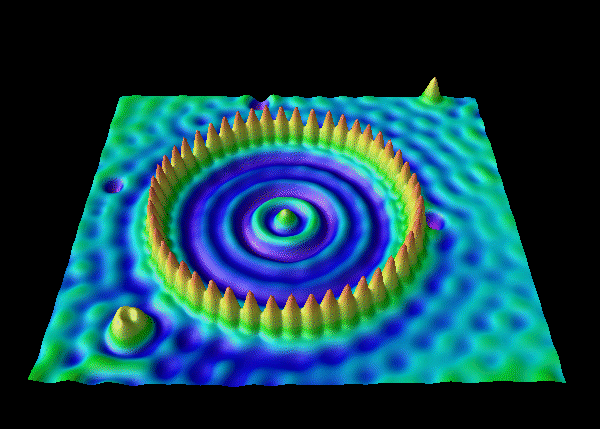
This exact probability distribution can be observed using an electron microscope. Below is a picture of a ring of iron atoms, whose probabilities are shown as peaks:
Degeneracy
It is possible for multiple pairs of quantum numbers to result in the same energy level. This situation is known as degeneracy and the energy levels are called degenerate.
For example, the energy state can be obtained with and or and . This is not as dramatic as, say, being obtained by and versus and . is said to have two-fold degeneracy, whereas has three-fold degeneracy (it can also be represented by and ).